Porphyrin functionalized Co(OH)2/GO nanocomposites as an excellent peroxidase mimic for colorimetric biosensing†
Abstract
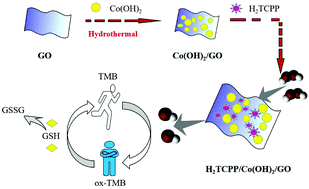
5,10,15,20-Tetrakis(4-carboxyl phenyl)porphyrin (Por) modified Co(OH)2 deposited on the surface of GO nanocomposites (Por/Co(OH)2/GO) were prepared and characterized by transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and XRD. For the first time, H2TCPP/Co(OH)2/GO is found to have enhanced peroxidase-like activity and catalyze the oxidation of the substrate 3,3,5,5-tetramethylbenzidine (TMB) by hydrogen peroxide (H2O2). Notably, the colorless TMB rapidly transformed into blue oxTMB in just 60 s, which was easily observed visually. The catalytic kinetics of H2TCPP/Co(OH)2/GO is in accord with the Michaelis–Menten equation. The catalytic mechanism of H2TCPP/Co(OH)2/GO nanocomposites is attributed to hydroxyl radicals (˙OH), due to decomposition of H2O2, which is verified by using terephthalic acid as a fluorescent probe. What's more, H2O2 can be detected in a wide linear detection range from 5 to 35 mM with a detection limit of 0.385 mM. Furthermore, based on the excellent peroxidase-like activity of H2TCPP/Co(OH)2/GO, a colorimetric sensor is established to sensitively detect glutathione (GSH) in a linear range from 10 to 300 μM with a low detection limit of 9.5 μM.


 Please wait while we load your content...
Please wait while we load your content...