Demonstration of a quantitative triplex LAMP assay with an improved probe-based readout for the detection of MRSA†
Abstract
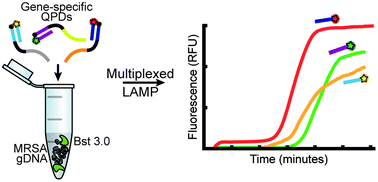
As molecular diagnostics move away from polymerase chain reaction (PCR) in order to target point-of-care testing applications, loop-mediated isothermal amplification (LAMP) is gaining popularity due its rapid, sensitive and specific detection with simpler instrumentation. However, while Taqman PCR enables real-time quantitative readout and multiplexed gene detection in single samples, analogous methods in LAMP are not yet broadly developed. To date, the real-time detection methods applied to LAMP involve turbidimetry or measuring fluorescence of an intercalator; however, both of these methods are nonspecific to the target of interest and do not allow for multiple gene detection in a single sample. Probe-based methods have been developed to address the need for specific target detection and multiplexed, one-pot reactions, but most of these methods have strict assay conditions and require the design of loop primers, which is not always possible. DARQ LAMP is a probe-based method that offers the most promise for quantitative and real-time multiplexed detection, as it has a relatively simple design and can be used in either a four-primer or six-primer system. However, previous work has only shown the assay to function well in a narrow range of reaction conditions, which is restrictive given that various LAMP assays require a broad range of conditions. In this work we investigate the use of the newest-generation strand-displacing polymerase and demonstrate that it has higher tolerance to reaction conditions than previous polymerases. Using the results from these studies, we demonstrate a single-reaction triplex assay for the detection of methicillin-resistant S. aureus (MRSA), which would not be possible with any of the previously reported LAMP systems.


 Please wait while we load your content...
Please wait while we load your content...