Dual-channel surface plasmon resonance monitoring of intracellular levels of the p53-MDM2 complex and caspase-3 induced by MDM2 antagonist Nutlin-3
Abstract
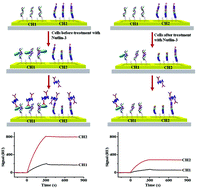
MDM2 can mediate the degradation of tumor suppressor p53 through an autoregulatory feedback loop, in which MDM2 abolishes wild-type p53 function and accelerates malignant transformation. However, the incorporation of MDM2 antagonist Nutlin-3 could reactivate the transcriptional activity of p53, up-regulate caspase-3, and induce apoptosis. In this work, the simultaneous and label-free monitoring of p53-MDM2 complex and caspase-3 levels in cancer cells before and after Nutlin-3 treatment was conducted using dual-channel surface plasmon resonance (SPR). The p53-MDM2 complex was captured in one fluidic channel covered with consensus double-stranded (ds)-DNA, while the other channel was pre-immobilized with caspase-3-specific biotinylated DEVD-containing peptides. To amplify the SPR signals, the attachment of streptavidin (SA)-conjugated anti-MDM2 antibody in both channels was achieved. The signal diversity before and after Nutlin-3 treatment is indicative of the difference in the levels of the intracellular p53-MDM2 complex and caspase-3. The limit of detection for p53-MDM2 and caspase-3 down to 4.54 pM and 0.03 ng mL−1, respectively, was attained. Upon treatment with Nutlin-3, MCF-7 cancer cells with wild-type p53 showed decreased expression of the p53-MDM2 complex and an increased caspase-3 level, while MDA-MB-231 cancer cells with mutant p53 exhibited an elevated caspase-3 level and unchanged p53-MDM2 complex expression. The apoptosis of MCF-7 and MDA-MB-231 cancer cells upon Nutlin-3 treatment follows a p53-dependent and a p53-independent pathway, respectively. The proposed method is sensitive, selective and label-free, holding great promise for assaying intracellular p53-MDM2 complex and caspase-3 levels and differentiating Nutlin-3-mediated p53-dependent or p53-independent apoptotic pathways.


 Please wait while we load your content...
Please wait while we load your content...