A possibly highly conducting state in an optically excited molecular crystal†
Abstract
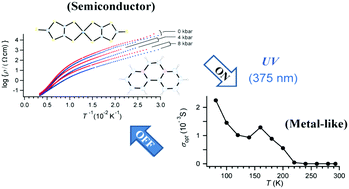
Unique electronic states and properties of the molecular charge transfer salts have been a center of interest for decades. A new molecular charge transfer salt, BPY[Ni(dmit)2]6·3CH3CN (BPY2+ = N,N′-ethylene 2,2′-bipyridinium, dmit2− = 2-thioxo-1,3-dithiole-4,5-dithiolato), exhibits semiconducting properties governed by its [Ni(dmit)2]− radical anions. This salt is an insulator but exhibits high conductance like a metallic substance. Structural, spectroscopic, and theoretical examinations reveal that the high conductance of the salt is based on a fluctuation in the charge distribution on the Ni(dmit)2 anions, which are closely and isotropically packed in a two-dimensional sheet owing to its Ni(dmit)2-rich stoichiometry (BPY : Ni(dmit)2 = 1 : 6). The qualitative conduction behavior is hardly affected by temperature and pressure unlike other organic charge transfer salts. However, UV irradiation (375 ± 5 nm), corresponding to a charge transfer between Ni(dmit)2 anions, perturbs the charge distribution to realize a highly conducting state based on our proposed analysis model.


 Please wait while we load your content...
Please wait while we load your content...