Deciphering the loss of persistent red luminescence in ZnGa2O4:Cr3+ upon Al3+ substitution†
Abstract
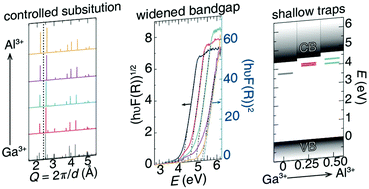
The development of persistent luminescent phosphors requires an improved understanding of the crystal structures and chemical compositions that generate their unique and distinctive optical properties. Currently, researchers generally agree that the long luminescent lifetimes arise from a relationship between the host crystal structure's bandgap and the location of defects, which form electron traps. Here, this notion is studied by preparing the red-emitting persistent luminescent phosphor solid solution Zn(Ga1−δAlδ)2O4:Cr3+ (δ = 0, 0.25, 0.50, 0.75, 1) using high-temperature, solid-state synthesis. The optical characterization of this series reveals that varying the main group metal in this spinel-type crystal structure causes a nearly linear change in the bandgap as a function of substitution. Interestingly, long luminescent lifetime measurements reveal a loss of persistent luminescence with increasing aluminum concentrations, even though there is an increase in the number of electron traps. Thermoluminescence measurements indicate shallower trap states are present with increasing aluminum concentration, which is conceivably the quenching mechanism. Analyzing the relationship between the change in composition and the loss of persistent luminescence reveals a threshold beyond which lattice defects will bring about the quenching of an observable long luminescent lifetime.


 Please wait while we load your content...
Please wait while we load your content...