Oxime-based 19-nortestosterone–pheophorbide a conjugate: bimodal controlled release concept for PDT†
Abstract
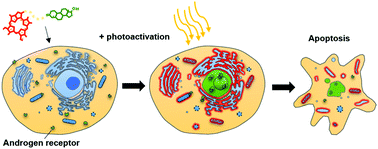
Photodynamic therapy has become a feasible direction for the treatment of both malignant and non-malignant diseases. It has been in the spotlight since FDA regulatory approval was granted to several photosensitizers worldwide. Nevertheless, there are still strong limitations in the targeting specificity that is vital to prevent systemic toxicity. Here, we report the synthesis and biological evaluation of a novel bimodal oxime conjugate composed of a photosensitizing drug, red-emitting pheophorbide a, and nandrolone (NT), a steroid specifically binding the androgen receptor (AR) commonly overexpressed in various tumors. We characterized the physico-chemical properties of the NT–pheophorbide a conjugate (NT–Pba) and singlet oxygen generation. Because light-triggered therapies have the potential to provide important advances in the treatment of hormone-sensitive cancer, the biological potential of this novel specifically-targeted photosensitizer was assessed in prostatic cancer cell lines in vitro using an AR-positive (LNCaP) and an AR-negative/positive cell line (PC-3). U-2 OS cells, both with and without stable AR expression, were used as a second cell line model. Interestingly, we found that the NT–Pba conjugate was not only photodynamically active and AR-specific, but also that its phototoxic effect was more pronounced compared to pristine pheophorbide a. We also examined the intracellular localization of NT–Pba. Live-cell fluorescence microscopy provided clear evidence that the NT–Pba conjugate localized in the endoplasmic reticulum and mitochondria. Moreover, we performed a competitive localization study with the excess of nonfluorescent NT, which was able to displace fluorescent NT–Pba from the cell interior, thereby further confirming the binding specificity. The oxime ether bond degradation was assayed in living cells by both real-time microscopy and a steroid receptor reporter assay using AR U-2 OS cells. Thus, NT–Pba is a promising candidate for both the selective targeting and eradication of AR-positive malignant cells by photodynamic therapy.


 Please wait while we load your content...
Please wait while we load your content...