Unimpeded migration of ions in carbon electrodes with bimodal pores at an ultralow temperature of −100 °C†
Abstract
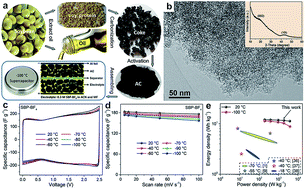
The ability to rapidly charge (and discharge) energy storage devices at extremely low temperature (down to −100 °C) is critical for low-temperature applications such as high altitude exploration and space missions. Electric double-layer supercapacitors (EDLCs) are promising energy storage devices under these conditions. However, it is still a great challenge to obtain EDLCs with both high gravimetric/volumetric capacitance and good rate performance at such low temperatures. We found that, in carbon-based EDLCs, the poor performance at low temperature was mainly caused by the sluggish desolvation of ions at the pore openings and low ion migration within pores. Further, we discovered that there exists a minimum pore opening size for ion adsorption and an effect of pore size on rate performance. These findings enable us to envisage a rational pore structure with a special bimodal distribution of micropores and mesopores. In this work, we successfully synthesized high surface area activated carbon (AC) with a similar structure. Based on this AC, record gravimetric/volumetric capacitance (173 F g−1 and 66 F cm−3 at 10 mV s−1 scan rate) and good rate performance (157 F g−1 and 60 F cm−3 at 100 mV s−1 scan rate) were obtained at a temperature of −100 °C.


 Please wait while we load your content...
Please wait while we load your content...