Electronic and optical properties of π-bridged perylenediimide derivatives: the role of π-bridges†
Abstract
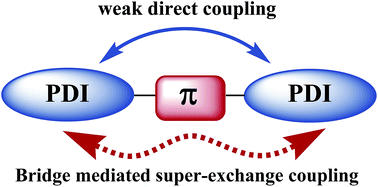
Constructing twisted molecular structures by linking multiple perylenediimide (PDI) units through a π-bridge has been recognized as an effective way to tune the active layer (phase separation) morphologies for PDI-based organic solar cells. But in the meantime, the introduced electron transfer between intramolecular PDI units will influence the exciton dissociation and electron transport properties of the devices. Herein, we have investigated the impact of π-bridge modes, including different bridge units and linking positions, on the electronic and optical properties of representative dimeric PDI derivatives by density functional theory (DFT) calculations. The calculated results point to that the intramolecular electron transfer is mainly controlled by super-exchange mechanism for the PDI–π–PDI dimeric derivatives. The super-exchange coupling is determined by the dihedral angle and energy level alignment between the π-bridge and PDI units as well as the electron density of the lowest unoccupied molecular orbital (LUMO) on the linkage site of PDI. Compared with the ortho position, the bay position of PDI possesses higher LUMO density and smaller dihedral angle when linking with the π-bridge, leading to stronger electronic coupling. Moreover, after the π-bridge is changed from benzene to triazine, the resultant smaller energy difference between the LUMOs of π-bridge and PDI will significantly enhance the super-exchange coupling. In addition, time-dependent DFT calculations show that the optical absorption spectra can be greatly modulated by the different π-bridge modes.


 Please wait while we load your content...
Please wait while we load your content...