New insights into the phase evolution in CuS during lithiation and delithiation processes†
Abstract
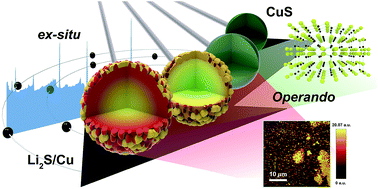
Materials that store lithium based on a conversion reaction are promising candidates for high-capacity electrodes for lithium-ion batteries. However, studies on the reaction mechanisms behind these electrochemical behaviors are still lacking, as it is difficult to detect the conversion products due to their nanoscale and structural disorder. Here, we report a study on the phase transformation of CuS during lithiation and delithiation processes using a combination of in situ and ex situ analytical tools. During the lithiation process, CuS exhibits two distinct voltage plateaus, undergoing an insertion reaction, followed by a conversion reaction into Li2S and Cu, without noticeable formation of Cu2−δS intermediates (0 ≤ δ < 1), which are discussed in the thermodynamic aspects on the basis of the operando and ex situ X-ray diffraction. Operando Raman spectroscopy and mapping reveals that the conversion reaction starts on the first plateau of lithiation, and the phase distribution of CuS changes on the microscale through the electrochemical process. It is believed that the conversion reaction competes with the insertion reaction that evolves a ‘Li-rich’ insertion phase until ∼0.75 V vs. Li/Li+. The high irreversibility of CuS upon initial lithiation/delithiation and the capacity fading during subsequent cycles appear to result from deep lithiation processes, including the conversion reaction, electrolyte decomposition and Cu nanoparticle agglomeration at low voltages, together with soluble polysulfide formation.
- This article is part of the themed collection: 2019 Journal of Materials Chemistry A HOT Papers


 Please wait while we load your content...
Please wait while we load your content...