Continuous desalination with a metal-free redox-mediator†
Abstract
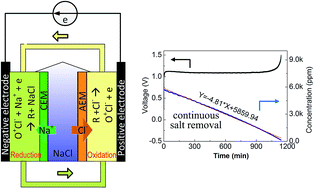
Deionization using electrochemical redox reactions has potentially been the latest desalination technique with high salt removal capacity and lower energy cost due to high capacity of electrode materials and electrochemical energy release. Herein, we presented a metal-free continuous electrodialysis desalination technology, which could directly desalt feed water at high concentrations to drinking water level during continuous treatment with unlimited salt removal capacity. The device consists of a recirculating 4-hydroxy-2,2,6,6-tetramethylpiperidine 1-oxyl (TEMPO) stream between the flowing positive and negative electrodes in series and the salt feed stream. During the charging process, the flowing TEMPO was oxidized at the positive reservoir, where the anion in the feed was captured through the anion exchange membrane, whereas the TEMPO recovery at the negative reservoir drove the cation removal in the feed through the cation exchange membrane by its reduction. Due to the redox reaction of TEMPO between the cyclical flow electrode streams, it acts as a “catalyst”, and the salt ions in the middle feed are continuously extracted. Our experiments demonstrate that feed water with a 5860 ppm salt concentration can be directly desalted to 136 ppm, a drinking water level, in a single batch treatment during the continuous charging process (salt removal of up to 97.7%). Furthermore, a three-membrane device was demonstrated to stop the salt concentration increase in the TEMPO electrode reservoirs; thus, fully continuous cycling of the TEMPO catalyst was realized without any salt change in the electrode reservoirs. Our research will provide a route for metal-free continuous desalination based on the molecular redox mediator of the electrode material and can be applied directly in open air environment and room temperature.


 Please wait while we load your content...
Please wait while we load your content...