Understanding the Li-ion storage mechanism in a carbon composited zinc sulfide electrode†
Abstract
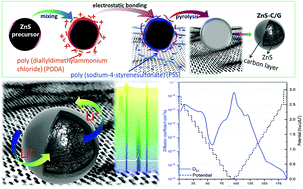
Sulfide compounds are interesting conversion electrode materials for Li-ion batteries, due to their high theoretical capacity. However, they suffer from large volumetric changes and fast capacity fading. To overcome these issues, nanosized zinc sulfide (ZnS) modified with polyelectrolytes and graphene (ZnS-C/G) has been synthesized and investigated as an enhanced conversion-alloying anode material. In situ synchrotron X-ray diffraction and X-ray absorption spectroscopy are used to elucidate the Li storage process during the 1st cycle. In addition, the evolution of internal resistance and the corresponding solid electrolyte interphase (SEI) formation during the 1st cycle are discussed based on electrochemical impedance spectroscopy and X-ray photoelectron spectroscopy. The results reveal that the formation of lithiated products and the SEI layer at different voltages can influence Li+ diffusion into the electrode. Moreover, an artificial carbon layer can not only facilitate Li+ transport but also avoid the direct formation of the SEI layer on the surface of active particles. Compared to bare ZnS, the ZnS-C/G electrode shows outstanding rate capability and cycling capacity (571 mA h g−1 after 120 cycles at a specific current of 1.0 A g−1 with a retention rate of 94.4%). The high capacity at elevated current density is ascribed to the contribution of capacitive charge storage.


 Please wait while we load your content...
Please wait while we load your content...