Boost-up electrochemical performance of MOFs via confined synthesis within nanoporous carbon matrices for supercapacitor and oxygen reduction reaction applications†
Abstract
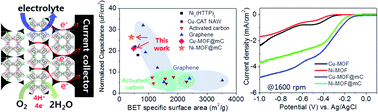
Utilization of metal–organic frameworks (MOFs) for electrochemical applications is significantly limited by their insulating nature and mechanical/chemical instability. One promising approach for the deployment of conventional MOFs in electrochemical applications is to fabricate MOF-based hybrids (MBHs) with conductive materials, which facilitate effective electron transfer via conductive additives between MOFs. Herein, we present a facile method for effective filling of Cu- and Ni-HKUST-1 (Hong Kong University of Science and Technology, and denoted hereafter as Cu-/Ni-MOF) inside 3D ordered mesoporous carbon (24 nm, mC), 3D ordered N-doped macroporous carbon (300 nm, NMC), and 3D ordered macroporous carbon (300 nm, MC), denoted as MOF@mC, MOF@NMC, and MOF@MC, respectively. The MOF@carbon matrix (MOF@CM) composites were intended for use as electrodes for electrical double layer capacitors (EDLCs) and as electrocatalysts for the oxygen reduction reaction (ORR). The EDLC performance of MOFs can be significantly improved by facilitating electron transfer through 3D conductive CM, reducing the electron pathway within the insulating MOF using a CM with small pores, and the choice of metal center with pronounced faradaic nature. Ni-MOF@mC exhibited superior specific surface area normalized areal capacitance (26.5 μF cm−2), exceeding most carbons and MOF-based EDLCs and outstanding long-term stability (91%@5000th). Furthermore, Cu-MOF@mC resulted in pronounced ORR activities, excellent methanol tolerance, and long-term stability. It is clearly demonstrated that conventional MOFs can be utilized for EDLCs and the ORR when conjugated with a 3D-connected nano-sized CM.


 Please wait while we load your content...
Please wait while we load your content...