An in situ anion exchange induced high-performance oxygen evolution reaction catalyst for the pH-near-neutral potassium borate electrolyte†
Abstract
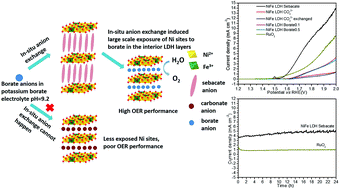
Research and development of highly efficient electrocatalysts for the kinetically sluggish oxygen evolution reaction (OER) under neutral or near-neutral pH conditions is urgent and challenging. Herein, for the first time we show that the anion exchange properties of sebacate anion intercalated NiFe layered double hydroxide (NiFe LDH Seb) can be taken advantage of to make NiFe LDHs a highly efficient OER catalyst for the pH-near-neutral potassium borate electrolyte (K-Bi, pH = 9.2). The NiFe LDH Seb nanosphere delivered a 1 mA cm−2 current density with an overpotential of 376 mV on a fluorine doped tin oxide (FTO) electrode in 0.1 M K-Bi (pH = 9.2) which is lower than that of RuO2 (393 mV) and all other reported earth-abundant OER catalysts on FTO electrodes. More importantly, a chronoamperometry test showed that for 24 hours, NiFe LDH Seb did not show any decay in its performance as an OER catalyst and always outperformed RuO2. To the best of our knowledge, this is the first time that anyone has reported a better performance of an earth-abundant OER catalyst than that of precious RuO2 in K-Bi (pH = 9.2). Besides, it also showed good OER performance under alkaline conditions. The excellent OER performance of NiFe LDH Seb was attributed to its large electrochemically active surface area and the in situ anion exchange induced large-scale exposure of Ni in the interior layers to the borate anions, which facilitates the activation of more Ni sites in the LDH. This work opens up a new path to develop LDH type OER catalysts for pH-neutral or pH-near-neutral electrolytes.


 Please wait while we load your content...
Please wait while we load your content...