In situ formation of highly controllable and stable Na3PS4 as a protective layer for Na metal anode†
Abstract
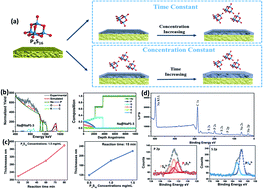
Na metal is considered as a promising anode material for Na metal batteries (NMBs) due to its high theoretical specific capacity and low electrochemical potential. However, the Na metal anode faces several challenges, including: (1) safety concerns related to dendrite formation; (2) low coulombic efficiency (CE) and poor cycling performance caused by the unstable solid electrolyte interphase (SEI); (3) an infinite volume change due to its hostless nature. In this paper, we demonstrate for the first time a facile and in situ solution-based method to synthesize an artificial protective layer of Na3PS4 (NaPS) on the surface of Na metal. Promisingly, the thickness and composition of the NaPS can be controlled through adjustment of the precursor concentration and reaction times. The thin amorphous NaPS layers on the surface of the Na metal will reduce parasitic side reactions with electrolyte components, increase the homogeneity of the SEI layer and improve the Na+ flux on the surface. Meanwhile, the ionically conductive NaPS can eliminate the strong electric field buildup at the surface, resulting in a more homogeneous Na plating/stripping process with suppressed Na dendrite growth. The design of the NaPS-coated Na anode opens up new opportunities for the realization of next-generation high-energy density Na metal batteries.


 Please wait while we load your content...
Please wait while we load your content...