Comparing water-mediated hydrogen-bonding in different polyelectrolyte complexes†
Abstract
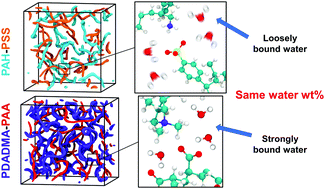
All-atom molecular dynamics simulations are used to investigate the polyelectrolyte-specific influence of hydration and temperature on water diffusion in hydrated polyelectrolyte complexes (PECs). Two model PECs were compared: poly(allylamine hydrochloride) (PAH)–poly(sodium 4-styrenesulfonate) (PSS) and poly(diallyldimethylammonium) (PDADMA)–poly(acrylic acid) (PAA). The findings show that the strength of the hydrogen bonding i.e. polyelectrolyte water interaction has enormous influence on the water mobility, which has implications for PEC structure and properties. A 10-fold difference in the average water diffusion coefficient between PAH–PSS and PDADMA–PAA PECs at the same hydration level is observed. The vast majority of the water molecules hydrating the PDADMA–PAA PECs, for hydrations in the range of 26–38 wt%, are effectively immobilized, whereas for PAH–PSS PECs the amount of immobilized water decreases with hydration. This points to the polyelectrolyte-specific character of the PE–water hydrogen bonding relationship with temperature. PAA–water hydrogen bonds are found to be significantly less sensitive to temperature than for PSS–water. The polyelectrolyte–water interactions, investigated via radial distribution function, hydrogen bond distance and angle distributions, are connected with resulting structure of the PECs. The PDADMA–PAA and PAH–PSS PECs are prepared experimentally and the states of water at different hydration levels is determined using differential scanning calorimetry (DSC). Experiments confirm the differences between PDADMA–PAA and PAH–PSS PECs observed in the theoretical modelling. The results suggest that the initial predictions of the PEC's bonding with water can be based on simple molecular-level considerations.


 Please wait while we load your content...
Please wait while we load your content...
