Charge transport and glassy dynamics in polymeric ionic liquids as reflected by their inter- and intramolecular interactions†
Abstract
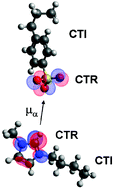
Polymeric ionic liquids (PILs) form a novel class of materials in which the extraordinary properties of ionic liquids (ILs) are combined with the mechanical stability of polymeric systems qualifying them for multifold applications. In the present study broadband dielectric spectroscopy (BDS), Fourier transform infrared spectroscopy (FTIR), AC-chip calorimetry (ACC) and differential scanning calorimetry (DSC) are combined in order to unravel the interplay between charge transport and glassy dynamics. Three low molecular weight ILs and their polymeric correspondents are studied with systematic variations of anions and cations. For all examined samples charge transport takes place by glassy dynamics assisted hopping conduction. In contrast to low molecular weight ILs the thermal activation of DC conductivity for the polymeric systems changes from a Vogel–Fulcher–Tammann- to an Arrhenius-dependence at a (sample specific) temperature Tσ0. This temperature has been widely discussed to coincide with the glass transition temperature Tg, a refined analysis, instead, reveals Tσ0 of all PILs under study at up to 80 K higher values. In effect, below the Tσ0 charge transport in PILs becomes more efficient – albeit on a much lower level compared to the low molecular weight pendants – indicating conduction paths along the polymer chain. This is corroborated by analysing the temperature dependence of specific IR-active vibrations showing at Tσ0 distinct changes in the spectral position and the oscillator strength, whereas other molecular units are not affected. This leads to the identification of charge transport responsive (CTR) as well as charge transport irresponsive (CTI) moieties and paves the way to a refined molecular understanding of electrical conduction in PILs.


 Please wait while we load your content...
Please wait while we load your content...