Crystallographic characterization of Y2C2n (2n = 82, 88–94): direct Y–Y bonding and cage-dependent cluster evolution†
Abstract
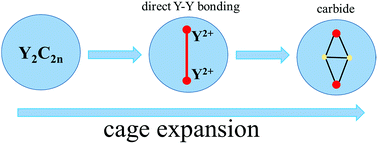
Direct yttrium–yttrium bonding has been a long-sought puzzle in organometallic chemistry to understand the catalytic processes that involve yttrium. Herein, we report the first crystallographic authentication of direct Y–Y bonding inside the hollow cavity of fullerene cages by forming endohedral metallofullerenes (EMFs). Based on an efficient separation/purification process, which involves Lewis-acid treatment and HPLC separation, we have obtained sufficient amounts of a series of Y2C2n (2n = 82, 88–94) isomers for systematic studies. The unambiguous single-crystal X-ray diffraction (XRD) crystallographic results show that two of them are di-EMFs, namely Y2@Cs(6)-C82 and Y2@C3v(8)-C82, in which the long-sought Y–Y single bond between the two divalent yttrium ions is experimentally confirmed for the first time. In contrast, all the other EMFs with relatively large cages are carbide cluster metallofullerenes (CCMFs), namely, Y2C2@Cs(15)-C86, Y2C2@C1(26)-C88, Y2C2@C2(41)-C90 and Y2C2@C2(61)-C92. Consistently, our computational results prove that these experimentally obtained EMFs are all abundant at the high temperatures for fullerene formation (∼1500–3000 K) due to the strong coordination ability of yttrium ions, which enables the formation of either direct Y–Y bonds (for Y2@Cs(6)-C82 and Y2@C3v(8)-C82) or the inclusion of a C2-unit (in Y2C2@Cs(15)-C86, Y2C2@C1(26)-C88, Y2C2@C2(41)-C90 and Y2C2@C2(61)-C92). Our results suggest that metal atoms such as yttrium tend to adopt a low valence state during the arc-discharge process because of the presence of the highly reductive carbon plasma in the chamber, enabling the formation of an Y2 dimer with direct Y–Y bonding in small cages like C82.
- This article is part of the themed collection: In celebration of Chinese New Year


 Please wait while we load your content...
Please wait while we load your content...