H-Bond donor parameters for cations†
Abstract
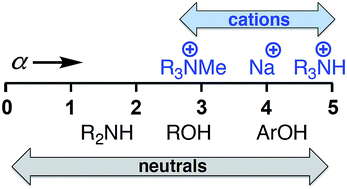
UV/Vis absorption and NMR spectroscopy titrations have been used to investigate the formation of complexes between cations and neutral H-bond acceptors in organic solvents. Complexes formed by two different H-bond acceptors with fifteen different cations were studied in acetone and in acetonitrile. The effects of water and ion pairing with the counter-anion were found to be negligible in the two polar solvents employed for this study. The data were used to determine self-consistent H-bond donor parameters (α) for a series of organic and inorganic cations; guanidinium, primary, tertiary and quaternary ammonium, imidazolium, methylpyridinium, lithium, sodium, potassium, rubidium and caesium. The results demonstrate the transferability of α parameters for cations between different solvents and different H-bond acceptor partners, allowing reliable prediction of cation recognition properties in different environments. Lithium and protonated nitrogen cations form the most stable complexes, but the α parameter is only 5.0, which is similar to the neutral H-bond donor 3-trifluoromethyl,4-nitrophenol (α = 5.1). Quaternary ammonium is the weakest H-bond donor investigated with an α value of 2.7, which is comparable to an alcohol. The α parameters for alkali metal cations decrease down the group from 5.0 (Li+) to 3.5 (Cs+).
- This article is part of the themed collection: Most popular 2018-2019 supramolecular chemistry articles


 Please wait while we load your content...
Please wait while we load your content...