Synthesis and reduction chemistry of mixed-Lewis-base-stabilised chloroborylenes†
Abstract
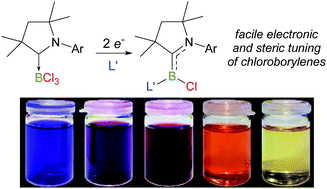
The one-electron reduction of (CAACMe)BCl3 (CAACMe = 1-(2,6-diisopropylphenyl)-3,3,5,5-tetramethylpyrrolidin-2-ylidene) yields the dichloroboryl radical [(CAACMe)BCl2]˙. Furthermore, the twofold reduction of (CAACMe)BCl3 in the presence of a range of Lewis bases (L = CAACMe, N-heterocyclic carbene, phosphine) yields a series of doubly base-supported (CAACMe)LBCl chloroborylenes, all of which were structurally characterised. NMR and UV-vis spectroscopic and electrochemical data for (CAACMe)LBCl show that the boron centre becomes more electron-rich and the HOMO–LUMO gap widens as L becomes less π-accepting. A [(CAACMe)BCl2]− boryl anion coordination polymer was isolated as a potential intermediate in these reductions. In most cases the reduction of the chloroborylenes resulted in the formation of the corresponding hydroborylenes or derivatives thereof, as well as ligand C–H activation products.


 Please wait while we load your content...
Please wait while we load your content...