Single-ion anisotropy and exchange coupling in cobalt(ii)-radical complexes: insights from magnetic and ab initio studies†
Abstract
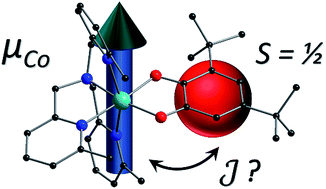
The concurrent effects of single-ion anisotropy and exchange interactions on the electronic structure and magnetization dynamics have been analyzed for a cobalt(II)-semiquinonate complex. Analogs containing diamagnetic catecholate and tropolonate ligands were employed for comparison of the magnetic behavior and zinc congeners assisted with the spectroscopic characterization and assessment of intermolecular interactions in the cobalt(II) compounds. Low temperature X-band (ν ≈ 9.4 GHz) and W-Band (ν ≈ 94 GHz) electron paramagnetic resonance spectroscopy and static and dynamic magnetic measurements have been used to elucidate the electronic structure of the high spin cobalt(II) ion in [Co(Me3tpa)(Br4cat)] (1; Me3tpa = tris[(6-methyl-2-pyridyl)methyl]amine, Br4cat2− = tetrabromocatecholate) and [Co(Me3tpa)(trop)](PF6) (2(PF6); trop− = tropolonate), which show slow relaxation of the magnetization in applied field. The cobalt(II)-semiquinonate exchange interaction in [Co(Me3tpa)(dbsq)](PF6)·tol (3(PF6)·tol; dbsq− = 3,5-di-tert-butylsemiquinonate, tol = toluene) has been determined using an anisotropic exchange Hamiltonian in conjunction with multistate restricted active space self-consistent field ab initio modeling and wavefunction analysis, with comparison to magnetic and inelastic neutron scattering data. Our results demonstrate dominant ferromagnetic exchange for 3+ that is of similar magnitude to the anisotropy parameters of the cobalt(II) ion and contains a significant contribution from spin–orbit coupling. The nature of the exchange coupling between octahedral high spin cobalt(II) and semiquinonate ligands is a longstanding question; answering this question for the specific case of 3+ has confirmed the considerable sensitivity of the exchange to the molecular structure. The methodology employed will be generally applicable for elucidating exchange coupling between orbitally-degenerate metal ions and radical ligands and relevant to the development of bistable molecules and their integration into devices.


 Please wait while we load your content...
Please wait while we load your content...