Guest recognition enhanced by lateral interactions†
Abstract
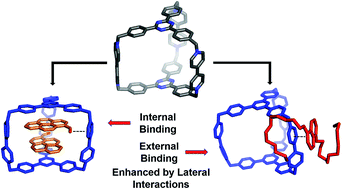
A hexacationic triangular covalent organic cage, AzaEx2Cage6+, has been synthesized by means of a tetrabutylammonium iodide-catalyzed SN2 reaction. The prismatic cage is composed of two triangular 2,4,6-triphenyl-1,3,5-triazine (TPT) platforms bridged face-to-face by three 4,4′-bipyridinium (BIPY2+) spacers. The rigidity of these building blocks leads to a shape-persistent cage cavity with an inter-platform distance of approximately 11.0 Å. This distance allows the cage to accommodate two aromatic guests, each of which is able to undergo π–π interactions with one of the two TPT platform simultaneously, in an A–D–D–A manner. In the previously reported prism-shaped cage, the spacers (pillars) are often considered passive or non-interactive. In the current system, the three BIPY2+ spacers are observed to play an important role in guest recognition. Firstly, the BIPY2+ spacers are able to interact with the carbonyl group in a pyrene-1-carbaldehyde (PCA) guest, by introducing lateral dipole–cation or dipole–dipole interactions. As a consequence, the binding affinity of the cage towards the PCA guest is significantly larger than that of pyrene as the guest, even although the latter is often considered to be a better π-electron donor. Secondly, in the case of the guest 1,5-bis[2-(2-(2-(2-hydroxyethoxy)ethoxy)ethoxy)ethoxy]naphthalene (BH4EN), the pillars can provide higher binding forces compared to the TPT platform. Hence, peripheral complexation occurs when AzaEx2Cage6+ accommodates BH4EN in MeCN. Thirdly, when both PCA and BH4EN are added into a solution of AzaEx2Cage6+, inclusion and peripheral complexation occur simultaneously to PCA and BH4EN respectively, even though the accommodation of the former guest seems to attenuate the external binding of the latter. This discovery of the importance of lateral interactions highlights the relationship between the electrostatic properties of a highly charged host and its complexation behavior, and as such, provides insight into the design of more complex hosts that bind guests in multiple locations and modes.
- This article is part of the themed collection: Most popular 2018-2019 supramolecular chemistry articles


 Please wait while we load your content...
Please wait while we load your content...