Bimetallic nickel-lutetium complexes: tuning the properties and catalytic hydrogenation activity of the Ni site by varying the Lu coordination environment†
Abstract
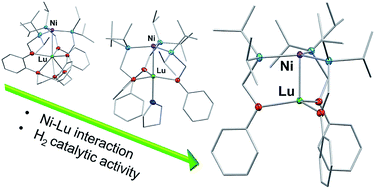
We present three heterobimetallic complexes containing an isostructural nickel center and a lutetium ion in varying coordination environments. The bidentate iPr2PCH2NHPh and nonadentate (iPr2PCH2NHAr)3tacn ligands were used to prepare the Lu metalloligands, Lu(iPr2PCH2NPh)3 (1) and Lu{(iPr2PCH2NAr)3tacn} (2), respectively. Reaction of Ni(COD)2 (where COD is 1,5-cyclooctadiene) and 1 afforded NiLu(iPr2PCH2NPh)3 (3), with a Lu coordination number (CN) of 4 and a Ni–Lu distance, d(Ni–Lu), of 2.4644(2) Å. Complex 3 can further bind THF to form 3-THF, increasing both the Lu CN to 5 and d(Ni–Lu) to 2.5989(4) Å. On the other hand, incorporation of Ni(0) into 2 provides NiLu{(iPr2PCH2NAr)3tacn} (4), in which the Lu coordination environment is more saturated (CN = 6), and the d(Ni–Lu) is substantially elongated at 2.9771(5) Å. Cyclic voltammetry of the three Ni–Lu complexes shows an overall ∼410 mV shift in the Ni(0/I) redox couple, suggesting tunability of the Ni electronics across the series. Computational studies reveal polarized bonding interactions between the Ni 3dz2 (major) and the Lu 5dz2 (minor) orbitals, where the percentage of Lu character increases in the order: 4 (6.0% Lu 5dz2) < 3-THF (8.5%) < 3 (9.3%). All three Ni–Lu complexes bind H2 at low temperatures (−30 to −80 °C) and are competent catalysts for styrene hydrogenation. Complex 3 outperforms 4 with a four-fold faster rate. Additionally, adding increasing THF equivalents to 3, which would favor build-up of 3-THF, decreases the rate. We propose that altering the coordination sphere of the Lu support can influence the resulting properties and catalytic activity of the active Ni(0) metal center.
- This article is part of the themed collection: Most popular 2019-2020 inorganic, main group and crystal engineering chemistry articles


 Please wait while we load your content...
Please wait while we load your content...