Selective recognition of choline phosphate by tripodal hexa-urea receptors with dual binding sites: crystal and solution evidence†
Abstract
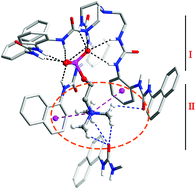
Two tripodal hexa-urea receptors functionalized with aromatic terminal groups are capable of binding choline phosphate (CP). Crystal structures of the host–guest complexes reveal that the zwitterion CP is efficiently encapsulated in the tripodal hosts in a dual-site binding mode. The phosphate tail of CP is coordinated by the urea groups and the quaternary ammonium head is bound in a ‘composite aromatic box’ through cation–π and hydrogen-bonding interactions. Such a partial aromatic binding environment for the Me3N–+ cation mimics that of most enzymes catalyzing the conversion of quaternary ammonium substrates. Moreover, NMR, ESI-MS, and fluorescence studies demonstrate the selective binding and sensing of CP over other competing species such as ADP, ATP, choline and derivatives.


 Please wait while we load your content...
Please wait while we load your content...