The nature of G⋯E–Y σ(3c–4e) in o-MenGCH2C6H4EY (MenG = Me2N and MeE; E = O, S, Se and Te; Y = F, Cl, Br, EMe and Me) with contributions from CT and compliance constants in noncovalent G⋯E interactions†
Abstract
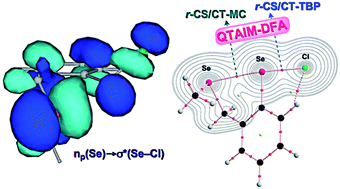
The intrinsic dynamic and static nature of G–*–E–*–Y σ(3c–4e) interactions was elucidated with the quantum theory of atoms in molecules dual functional analysis (QTAIM-DFA), employing o-MenGCH2C6H4EY (MenG = Me2N and MeE; E = O, S, Se and Te; Y = F, Cl, Br, I, EMe and Me). Asterisks (*) are employed to emphasize the existence of bond critical points (BCPs) on the bond paths (BPs), corresponding to the interactions in question. Data from the fully optimized structure correspond to the static nature of interactions. The dynamic nature is called the intrinsic dynamic nature if the perturbed structures are generated using the coordinates derived from the compliance constants. Basis sets of the Sapporo-TZP type with diffusion functions are employed for the heteroatoms at the MP2 level. The noncovalent G–*–E interactions in GEY σ(3c–4e) are predicted to demonstrate van der Waals bonding to CT-TBP (trigonal bipyramidal adduct formation through charge transfer) nature, while the E–*–Y bonds have the covalent nature. Some E–F bonds show strong ionic character when G–*–E is predicted to be stronger than E–*–Y. The contributions of the CT terms to the G–*–E interactions, evaluated with NBO, are discussed in relation to the predicted nature. The E(2) values based on NBO are strongly correlated to the compliance constants for the G–*–E interactions if suitably treated separately.


 Please wait while we load your content...
Please wait while we load your content...