Promoted dispersion and uniformity of active species on Fe–Ce–Al catalysts for efficient NO abatement†
Abstract
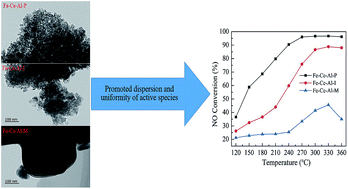
Fe–Ce–Al catalysts were synthesized by the co-precipitation method (labeled as Fe–Ce–Al–P), co-impregnation method (Fe–Ce–Al–I), and direct mixing method (Fe–Ce–Al–M), respectively, and used for effective removal of NO. The synthesized catalysts were characterized by many methods including N2 physisorption, X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), NH3-temperature programmed desorption (NH3-TPD), H2-temperature programmed reduction (H2-TPR), high-resolution transmission electron microscopy (HR-TEM), and energy dispersive spectroscopy (EDS) mapping. The results show that the synthesis methods greatly influence the catalytic performance of catalysts. The Fe–Ce–Al–P catalyst prepared by the co-precipitation method yields the highest catalytic performance, while the Fe–Ce–Al–I and Fe–Ce–Al–M catalysts exhibit relatively low catalytic activity. The co-precipitation method can promote the accumulation and dispersion of more surface active species on the catalyst surface, and provide smaller particle size of active species and generate more uniform particle size distribution, while these characteristics can't be obtained by the co-impregnation method and direct mixing method. Moreover, the co-precipitation method could produce the highest surface area and enhanced redox ability and surface acidity of the catalyst, which resulted from the high dispersion and uniform distribution of surface active species. These may be the key factors to the superior catalytic performance of the Fe–Ce–Al–P catalyst.


 Please wait while we load your content...
Please wait while we load your content...