Concise total syntheses of phelligridins A, C, and D†
Abstract
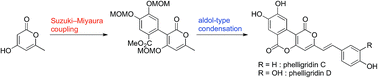
We have established a concise and scalable synthetic pathway for phelligridins A (1), C (2) and D (3). The synthetic highlights were Suzuki–Miyaura coupling and aldol-type condensation of α-pyrone. Phelligridin A was synthesized in four steps, while phelligridins C and D were each synthesized in six steps. Furthermore, we have revealed that the whole structure is essential for the cytotoxicity of phelligridins.


 Please wait while we load your content...
Please wait while we load your content...