Moderate NaNO2 etching enables easy crystallinity optimization of g-C3N4 with superior photoreduction performance
Abstract
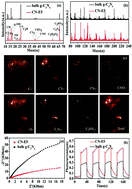
As a promising visible-light-responsive catalyst, graphitic carbon nitride (g-C3N4) has been widely used in environmental and energy applications owing to its unique semiconducting optoelectronic properties. However, the photocatalytic performance of bulk g-C3N4 synthesized using N-containing precursors via a conventional thermal polycondensation process is generally limited by low crystallinity caused by incomplete polymerization. In addition to forced polymerization under high-pressure conditions, removing the unpolymerized and inactive part of bulk g-C3N4 through controllable etching is another practical way to optimize its crystallinity. Therefore, we developed an economical and general method to fabricate g-C3N4 with high crystallinity and excellent photocatalytic properties, in which cheap sodium nitrite aqueous solution is utilized as a moderate etching agent. The compositions and physiochemical properties of the products were comprehensively investigated by X-ray diffraction, Fourier-transform infrared spectroscopy, ultraviolet visible diffuse reflectance spectroscopy, X-ray photoelectron spectroscopy, N2 adsorption–desorption, and electrochemical impedance spectroscopy measurements. In addition, ToF-SIMS (time-of-flight secondary ion mass spectrometry), a new surface analysis technology, was also used to determine the surface and structural characteristics of all samples. The obtained g-C3N4 shows ideal high-crystallinity features and excellent charge transfer ability, leading to significantly improved Cr(VI) reduction activity under visible light. Moreover, there is only a low content of nitrite residue (63.3 mg g−1) in the final g-C3N4 product, indicating that the method employing NaNO2, which is widely used in the food industry, is safe and waste-free. Therefore, the new controllable NaNO2 etching method is cleaner and more efficient than commonly-used corrosion approaches based on strong acids or strong oxidants, and thus affords a new strategy in the regulation and structural design of various catalysts.


 Please wait while we load your content...
Please wait while we load your content...