Directed motion of water droplets on multi-gradient photopolymer surfaces†
Abstract
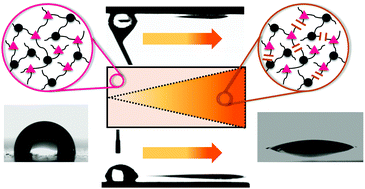
The present work highlights the controlled directional movement of water droplets across a photopolymer surface. The movement is propelled by multi-gradients including a wettability gradient and a Laplace pressure gradient. Both gradients are conveniently adjusted by light employing a photoresponsive thiol–yne photopolymer. o-Nitrobenzyl alcohol derivatives with terminal alkyne groups are synthetized and cured across di- and tri-functional thiols upon visible light exposure. The wettability gradient is generated in a subsequent step involving an asymmetrical irradiation of the polymer surface with light in the UV-A spectral region. Polar groups are formed in the exposed areas due to the photocleavage of the chromophore and photo-oxidation reactions (upon prolonged UV exposure in air). The wettability rises with increasing exposure dose and gradient surfaces are prepared with static water contact angles ranging from 97 to 19°. By simultaneously inscribing the wettability gradient in wedge-shaped patterns, a Laplace pressure gradient is realized on the photopolymer surface, which can be easily tailored by the size and the angle of the wedge. The combination of both gradients enables a rapid and directed movement of water droplets (2 μL droplet) over a reasonable distance (up to 10 mm). Due to the high adhesion of the photopolymer surface, the droplet can be driven in a controlled way, even if the surface is inclined (20°) or turned upside down.


 Please wait while we load your content...
Please wait while we load your content...