Identification of crucial bottlenecks in engineered polyketide biosynthesis†
Abstract
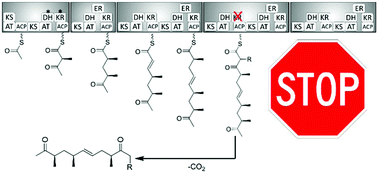
The concept of combinatorial biosynthesis promises access to compound libraries based on privileged natural scaffolds. Ever since the elucidation of the biosynthetic pathway towards the antibiotic erythromycin A in 1990, the predictable manipulation of type I polyketide synthase megaenzymes was investigated. However, this goal was rarely reached beyond simplified model systems. In this study, we identify the intermediates in the biosynthesis of the polyether monensin and numerous mutated variants using a targeted metabolomics approach. We investigate the biosynthetic flow of intermediates and use the experimental setup to reveal the presence of selectivity filters in polyketide synthases. These obstruct the processing of non-native intermediates in the enzymatic assembly line. Thereby we question the concept of a truly modular organization of polyketide synthases and highlight obstacles in substrate channeling along the cascade. In the search for the molecular origin of a selectivity filter, we investigate the role of different thioesterases in the monensin gene cluster and the connection between ketosynthase sequence motifs and incoming substrate structures. Furthermore, we demonstrate that the selectivity filters do not apply to new-to-nature side-chains in nascent polyketides, showing that the acceptance of these is not generally limited by downstream modules.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...