Synthesis of (difluoromethyl)naphthalenes using the ring construction strategy: C–C bond formation on the central carbon of 1,1-difluoroallenes via Pd-catalyzed insertion†
Abstract
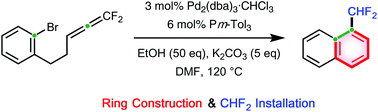
The insertion of 1,1-difluoroallenes was carried out to form a C–C bond exclusively on their central carbon. o-Bromophenyl-bearing 1,1-difluoroallenes underwent intramolecular insertion in the presence of a palladium catalyst. Regioselective C–C bond formation occurred to form a six-membered carbocycle, leading to pharmaceutically and agrochemically promising difluoromethylated naphthalenes.
- This article is part of the themed collections: Synthetic methodology in OBC and Fluorine Chemistry


 Please wait while we load your content...
Please wait while we load your content...