DANPY (dimethylaminonaphthylpyridinium): an economical and biocompatible fluorophore†
Abstract
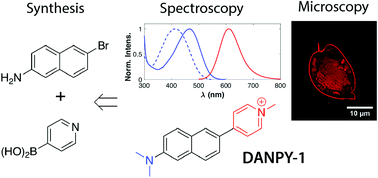
Dyes with nonlinear optical (NLO) properties enable new imaging techniques and photonic systems. We have developed a dye (DANPY-1) for photonics applications in biological substrates such as nucleic acids; however, the design specification also enables it to be used for visualizing biomolecules. It is a prototype dye demonstrating a water-soluble, NLO-active fluorophore with high photostability, a large Stokes shift, and a favorable toxicity profile. A practical and scalable synthetic route to DANPY salts has been optimized featuring: (1) convergent Pd-catalyzed Suzuki coupling with pyridine 4-boronic acid, (2) site-selective pyridyl N-methylation, and (3) direct recovery of crystalline intermediates without chromatography. We characterize the optical properties, biocompatibility, and biological staining behavior of DANPY-1. In addition to stability and solubility across a range of polar media, the DANPY-1 chromophore shows a first hyperpolarizability similar to common NLO dyes such as Disperse Red 1 and DAST, a large two-photon absorption cross section for its size, substantial affinity to nucleic acids in vitro, an ability to stain a variety of cellular components, and strong sensitivity of its fluorescence properties to its dielectric environment.


 Please wait while we load your content...
Please wait while we load your content...