Synthesis of hydrophobic insulin-based peptides using a helping hand strategy†
Abstract
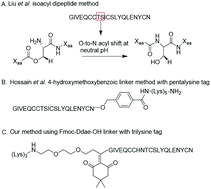
The introduction of solid-phase peptide synthesis in the 1960s improved the chemical synthesis of both the A- and B-chains of insulin and insulin analogs. However, the subsequent elaboration of the synthetic peptides to generate active hormones continues to be difficult and complex due in part to the hydrophobicity of the A-chain. Over the past decade, several groups have developed different methods to enhance A-chain solubility. Two of the most popular methods are use of isoacyl dipeptides, and the attachment of an A-chain C-terminal pentalysine tag with a base-labile 4-hydroxymethylbenzoic acid linker. These methods have proven effective but can be limited in scope depending on the peptide sequence of a specific insulin. Herein we describe an auxiliary approach to enhance the solubility of insulin-based peptides by incorporating a tri-lysine tag attached to a cleavable Fmoc-Ddae-OH linker. Incorporation of this linker, or “helping hand”, on the N-terminus greatly improved the solubility of chicken insulin A-chain, which is analogous to human insulin, and allowed for coupling of the insulin A- and B-chain via directed disulfide bond formation. After formation of the insulin heterodimer, the linker and tag could be easily removed using a hydrazine buffer (pH 7.5) to obtain an overall 12.6% yield based on A-chain. This strategy offers an efficient method to enhance the solubility of hydrophobic insulin-based peptides as well as other traditionally difficult peptides.
- This article is part of the themed collection: New Talent


 Please wait while we load your content...
Please wait while we load your content...