Nanomechanical sub-surface mapping of living biological cells by force microscopy†
Abstract
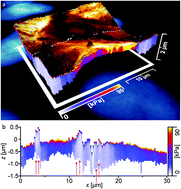
Atomic force microscopy allows for the nanomechanical surface characterization of a multitude of types of materials with highest spatial precision in various relevant environments. In recent years, researchers have refined this methodology to analyze living biological materials in vitro. The atomic force microscope thus has become an essential instrument for the (in many cases) non-destructive, high-resolution imaging of cells and visualization of their dynamic mechanical processes. Mapping force versus distance curves and the local evaluation of soft samples allow the operator to “see” beneath the sample surface and to capture the local mechanical properties. In this work, we combine atomic force microscopy with fluorescence microscopy to investigate cancerous epithelial breast cells in culture medium. With unprecedented spatial resolution, we provide tomographic images for the local elasticity of confluent layers of cells. For these particular samples, a layer of higher elastic modulus located directly beneath the cell membrane in comparison with the average elastic properties was observed. Strikingly, this layer appears to be perforated at unique locations of the sample surface of weakest mechanical properties where distinct features were visible permitting the tip to indent farthest into the cell's volume. We interpret this layer as the cell membrane mechanically supported by the components of the cytoskeleton that is populated with sites of integral membrane proteins. These proteins act as breaking points for the indenter thus explaining the mechanical weakness at these locations. In contrast, the highest mechanical strength of the cell was found at locations of the cell cores as cross-checked by fluorescence microscopy images of staining experiments, in particular at nucleoli sites as the cumulative elastic modulus there comprises cytoskeletal features and the tight packing ribosomal DNA of the cell.


 Please wait while we load your content...
Please wait while we load your content...