Self-assembly of a robust hydrogen-bonded octylphosphonate network on cesium lead bromide perovskite nanocrystals for light-emitting diodes†
Abstract
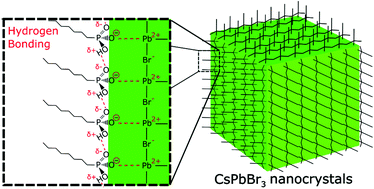
We report the self-assembly of an extensive inter-ligand hydrogen-bonding network of octylphosphonates on the surface of cesium lead bromide nanocrystals (CsPbBr3 NCs). The post-synthetic addition of octylphosphonic acid to oleic acid/oleylamine-capped CsPbBr3 NCs promoted the attachment of octylphosphonate to the NC surface, while the remaining oleylammonium ligands maintained the high dispersability of the NCs in non-polar solvent. Through powerful 2D solid-state 31P–1H NMR, we demonstrated that an ethyl acetate/acetonitrile purification regime was crucial for initiating the self-assembly of extensive octylphosphonate chains. Octylphosphonate ligands were found to preferentially bind in a monodentate mode through P–O−, leaving polar P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and P–OH groups free to form inter-ligand hydrogen bonds. The octylphosphonate ligand network strongly passivated the nanocrystal surface, yielding a fully-purified CsPbBr3 NC ink with PLQY of 62%, over 3 times higher than untreated NCs. We translated this to LED devices, achieving maximum external quantum efficiency and luminance of 7.74% and 1022 cd m−2 with OPA treatment, as opposed to 3.59% and 229 cd m−2 for untreated CsPbBr3 NCs. This represents one of the highest efficiency LEDs obtained for all-inorganic CsPbBr3 NCs, accomplished through simple, effective passivation and purification processes. The robust binding of octylphosphonates to the perovskite lattice, and specifically their ability to interlink through hydrogen bonding, offers a promising passivation approach which could potentially be beneficial across a breadth of halide perovskite optoelectronic applications.
O and P–OH groups free to form inter-ligand hydrogen bonds. The octylphosphonate ligand network strongly passivated the nanocrystal surface, yielding a fully-purified CsPbBr3 NC ink with PLQY of 62%, over 3 times higher than untreated NCs. We translated this to LED devices, achieving maximum external quantum efficiency and luminance of 7.74% and 1022 cd m−2 with OPA treatment, as opposed to 3.59% and 229 cd m−2 for untreated CsPbBr3 NCs. This represents one of the highest efficiency LEDs obtained for all-inorganic CsPbBr3 NCs, accomplished through simple, effective passivation and purification processes. The robust binding of octylphosphonates to the perovskite lattice, and specifically their ability to interlink through hydrogen bonding, offers a promising passivation approach which could potentially be beneficial across a breadth of halide perovskite optoelectronic applications.


 Please wait while we load your content...
Please wait while we load your content...