In situ construction of hollow carbon spheres with N, Co, and Fe co-doping as electrochemical sensors for simultaneous determination of dihydroxybenzene isomers†
Abstract
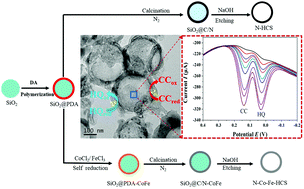
Control of the active sites/centers plays an important role in the design of novel electrode materials with unusual properties and achievement of sensors with high performance. In this study, three-dimensional (3D) freestanding multi-doped hollow carbon spheres (N–Co–Fe–HCS) with a layer thickness of 30 nm, which contained multiple active sites of the heteroatom N and transition metals (Co and Fe), were synthesized via a simple template method (with SiO2 as the template) and cost-efficient in situ self-polymerization, self-adsorption/reduction and carbonization strategies. Moreover, a series of hollow carbon sphere composites of the same family (N–HCS, N–Co–HCS and N–Fe–HCS) were prepared by this sensible process using the same method and precursors but different doping elements. These differences lead to different active sites/centers from hollow carbon spheres and improved electrocatalytic activities for dihydroxybenzene isomers. Furthermore, N–Co–Fe–HCS as an electrochemical sensor exhibited excellent simultaneous qualitative and quantitative determination performance for catechol (CC) and hydroquinone (HQ). The detection limit and the linear range were 75 nmol L−1 and 0.5–500 μmol L−1 for CC and 80 nmol L−1 and 0.5–1500 μmol L−1 for HQ, respectively. The interference from the components coexisting in river water on the detection of CC and HQ was not observed. These results indicate that high-performance electrochemical sensors can be constructed by in situ multi-element doping into electrode materials to achieve multi-active sites.


 Please wait while we load your content...
Please wait while we load your content...