Tracking sodium migration in TiS2 using in situ TEM†
Abstract
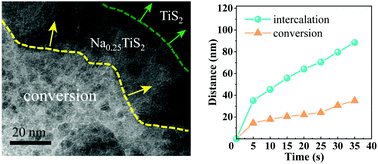
For alkali-metal ion batteries, revealing the phase transformation and the ion migration dynamics in the electrodes is vital for understanding how the electrodes work and thereby how we can improve them. Here, using in situ transmission electron microscopy, we track the structural evolution and migration dynamics during sodium insertion into TiS2 nanostructures with the lattice fringe resolution. We find that the sodiation process of TiS2 is initiated by an intercalation reaction and followed by a conversion reaction. From the same reaction event, the velocity of intercalation/conversion phase boundary migration is found to be ∼1.0–1.7 nm s−1, while the pristine/intercalation phase boundary migrates at a velocity of ∼2.5 nm s−1. The sodium migration leads to structural fracture to form nanometer-sized domains (∼3 nm) with volume expansion. During migration, Na prefers to transport along specific directions. Furthermore, a superstructured Na0.25TiS2 intermediate phase with ordered Na ions occupied within the (0001) plane is formed at the reaction front, which is different from the common staging phase. These findings help us understand the working principle and the failure mechanism of the sodium ion battery and also provide useful insights into the general ionic doping of transition metal dichalcogenides.


 Please wait while we load your content...
Please wait while we load your content...