Tuning the structure of bifunctional Pt/SmMn2O5 interfaces for promoted low-temperature CO oxidation activity†
Abstract
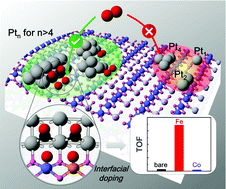
The interfacial structure of metal–oxide composite catalysts plays a vital role in heterogeneous catalysis, which is crucial to the adsorption and activation of reactants. Herein, the interfacial effects of bare and Fe/Co/Ni doped SmMn2O5 mullite oxide supported Pt clusters on CO oxidation have been investigated by first-principles based microkinetics analysis. A robust formation of Pt/Mn2 trimer structures is demonstrated at the bifunctional interfaces irrespective of the Ptn cluster's size, which can provide spatially separated sites for CO adsorption and O2 dissociation. The binding strength of CO at the interfacial Pt sites is in the optimal range due to the charge transfer from Pt clusters to oxide, while the strong polarization of Mn2 dimers induced by Pt clusters with stable three-dimensional morphologies can lower the energy barrier of O2 dissociation. Based on the microkinetics analysis, the O2 dissociation is the rate-determining step in the full CO oxidation cycle, and the introduction of Mn–Fe hetero-dimers at the interface is predicted to further enhance the low temperature CO oxidation activity of Pt/SmMn2O5 catalysts.


 Please wait while we load your content...
Please wait while we load your content...