Eight out of eight: a detailed kinetic study on the reactivities of the eight hydroxyl groups of sucrose with phenyl isocyanate†
Abstract
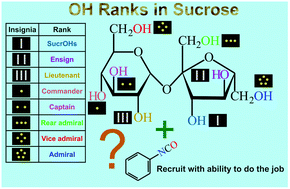
In this paper a detailed kinetic study between sucrose (S) and phenyl isocyanate (PI) in dimethylsulfoxide is reported for the first-time. In order to obtain monosubstituted sucrose derivatives overwhelmingly, i.e., when only one phenyl isocyanate molecule is attached to sucrose, sucrose was applied in a high molar excess to isocyanate (the molar ratio of S/PI was 16/1). Such an experimental setup indeed resulted in the formation of eight different sucrose derivatives which were separated and detected by high performance liquid chromatography combined with UV spectroscopy and mass spectrometry (HPLC-UV/MS). Under these experimental conditions, the disubstituted sucrose derivatives formed could be detected only in a few percent (ca. 2% with respect to the monosubstituted sucrose derivatives). Furthermore, by means of HPLC-UV/MS, HPLC-UV/MS/MS and NMR methods such as 1H-, 13C-NMR and 2D-NMR the reactivity of each of the eight OH groups of sucrose was determined. It was found that the most reactive moieties of sucrose are the three primary OH groups (hydroxymethyl) and the reactivity of the sucrose's OH groups decreases in the order of k(OH)6′(1) > k(OH)6(0.84) > k(OH)1′(0.31) > k(OH)3(0.25) > k(OH)4(0.23) > k(OH)2(0.13) > k(OH)4′(0.11) > k(OH)3′(0.09), where the numbers in subscript and in brackets represent the OH position in sucrose and the relative reactivity with respect to that of the highest one. The reactivity order is in line with the Mulliken charges of the OH groups of sucrose calculated by density functional theory (DFT) and also with those expected according to the theory of chromatographic separation. The dependences of the relative reactivities on temperature were also investigated and the differences between the corresponding activation parameters were also determined and discussed.


 Please wait while we load your content...
Please wait while we load your content...