The promoter effect of Co on the catalytic activity of the Cu oxide active phase supported on Al2O3 in the hydrogenolysis of glycerol†
Abstract
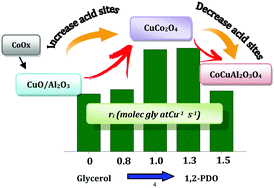
The promoting effect of cobalt oxide on alumina-supported Cu oxide catalysts for the hydrogenolysis of glycerol was investigated. A series of Co(y)Cu/Al2O3 oxide catalysts with a fixed Cu loading of 7 wt% and a variable Co loading from 0 to 1.5 wt% was prepared by successive wet impregnation. The catalysts were characterized by X-ray diffraction (XRD), nitrogen sorption, temperature programmed reduction (TPR), ultraviolet-diffuse reflectance spectroscopy (UV-DRS), temperature programmed desorption of ammonia (TPD-NH3), and X-ray photoelectron spectroscopy (XPS). The catalysts were tested in a batch reactor at 220 °C and 5 MPa of H2. The activity of the catalysts on a per mole of Cu basis increased with Co content up to the plateau, and subsequently decreased at higher Co content. The increase of activity is attributed to the combined effect of the distortion of the octahedral geometry of CuO by the presence of CoO and the increase in the number of surface acid sites. In contrast, the subsequent decrease of activity at higher loading is related to the formation of less reducible, less acidic, and less active aluminates. The selectivity of products did not vary substantially with Co loading, indicative of the existence of active sites of similar characteristics on all the catalysts. Overall, all the catalysts favored the route comprising of C–O hydrogenolysis followed by hydrogenation, at the expense of the pathway involving C–C hydrogenolysis, leading to the predominant formation of 1,2-propanediol (1,2-PDO). This is probably due to the prevalence of metallic active sites over acidic sites on the surface, ensuring that partial removal of oxygen occurred without the loss of carbon atoms.


 Please wait while we load your content...
Please wait while we load your content...