Secondary compounds in the catalytic hydrogenation of enone and allylic alcohol prostaglandin intermediates: isolation, characterization, and X-ray crystallography†‡
Abstract
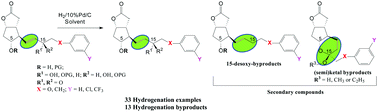
The discovery of the intraocular pressure reduction of 13,14-hydrogenated prostaglandins, among which Latanoprost is the most used drug in glaucoma treatment, has stimulated researchers to improve the synthesis of prostaglandins and their structural analogues. We studied the hydrogenation of the 13,14-double bond of unsaturated allylic and ketone γ-lactone prostaglandin intermediates with a 17-phenyl or 16-(3-chloro- or 3-trifluoromethyl)phenoxy radical in the ω-side chain; these intermediates, without α-side chains, are the last in the Corey prostaglandin sequence that can be chemo-selectively hydrogenated. The influence of protection of the hydroxyl(s) on the yield and the formation of secondary compounds in the hydrogenation were also studied. Moreover, 33 hydrogenation reactions were performed, and 13 secondary compounds were isolated and fully characterized; two compounds were also characterized by X-ray crystallography, all done to understand the specified hydrogenation reactions. A reduction by a factor of ten in the quantity of the noble palladium catalyst needed has also been achieved. The amount of catalyst, solvent, and reaction time of the hydrogenations were also described. The data for these compounds are not found in the literature, and our results may be useful not only for researchers in the field, but also for organic chemists who encounter secondary products in this type of hydrogenation.


 Please wait while we load your content...
Please wait while we load your content...