The catalytic evaluation of bimetallic Pd-based nanocatalysts supported on ion exchange resin in nitro and alkyne reduction reactions†
Abstract
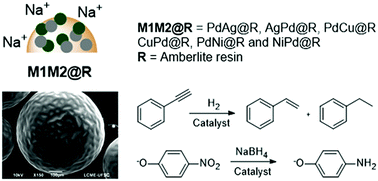
Deposition on the surface of amberlite resin was conducted to produce two different types of bimetallic nanoparticles, combining one of three different metals (M = Cu, Ag and Ni) with Pd (M–Pd@R and Pd–M@R). To obtain the bimetallic nanoparticles, the four different metal ions were firstly immobilized on the resin surface by ion exchange and monometallic nanoparticles were formed by reduction of the metal ions with NaBH4. By changing the sequence of metal ion immobilization on the resin, six bimetallic supported catalysts were prepared. Surface oxidation of the metals on the surface layer is inhibited in some cases and the catalytic propensity for certain reactions is improved. After careful characterization by scanning and transmission electron microscopy, X-ray photoelectron spectroscopy and X-ray absorption near edge structure, the activity of the catalysts was evaluated performing kinetic studies on the reduction reactions of 4-nitrophenol and phenylacetylene. The kinetic study revealed that the geometric factor, opening up catalyst sites through the ensemble effect, seems to be the main reason for the differentiated catalytic activity of the nanoparticle composites. Moreover, high selectivity for styrene in the hydrogenation of phenylacetylene was observed for Cu-containing catalysts, with almost total conversion of the substrate.


 Please wait while we load your content...
Please wait while we load your content...