Ion diffusion across a disorder-to-order phase transition in a poly(ethylene oxide)-b-poly(silsesquioxane) block copolymer electrolyte†
Abstract
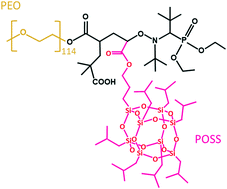
Nanostructured block copolymer electrolytes composed of organic and inorganic moieties have the potential to enable solid-state batteries. Practical uses of these materials, however, require an understanding of the microscopic and macroscopic ion transport properties across the microphase-separated systems. The self-diffusion of salt ions across a disorder-lamellar phase transition in a nanostructured poly(ethylene oxide)-b-poly(silsesquioxane) copolymer was studied using pulsed-field gradient NMR (PFG-NMR) and changes in the morphology were studied using small-angle X-ray scattering. The diffusion of the salt is isotropic when the polymer electrolyte is disordered and locally anisotropic when the polymer is microphase separated. The difference between the diffusion coefficient parallel to the lamellae, D∥, and the diffusion coefficient perpendicular to the lamellae, D⊥, measured using PFG-NMR, increases with increasing segregation strength. The anisotropy of diffusion parallels changes in the morphology measured by small-angle X-ray scattering.
- This article is part of the themed collection: Charge Transporting Nanostructured Polymers for Electrochemical Systems


 Please wait while we load your content...
Please wait while we load your content...