Functionalised tetrahydrofuran fragments from carbohydrates or sugar beet pulp biomass†
Abstract
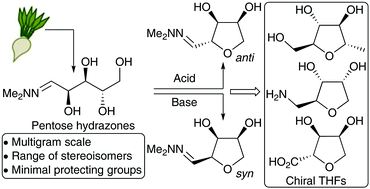
Carbohydrate biomass represents a potentially valuable sustainable source of raw materials for chemical synthesis, but for many applications, selective deoxygenation/dehydration of the sugars present is necessary to access compounds with useful chemical and physical properties. Selective dehydration of pentose sugars to give tetrahydrofurans can be achieved by treatment of the corresponding N,N-dimethylhydrazones under acidic or basic conditions, with the two approaches showing complementary stereoselectivity. The dehydration process is readily scalable and the THF hydrazones derived from arabinose, ribose, xylose and rhamnose were converted into a range of useful fragments containing primary alcohol, ketone, carboxylic acid or amine functional groups. These compounds have potentially useful physiochemical properties making them suitable for incorporation into fragment/lead generation libraries for medicinal chemistry. It was also shown that L-arabinose hydrazone could be obtained selectively from a crude sample of hydrolysed sugar beet pulp.


 Please wait while we load your content...
Please wait while we load your content...