Leveraging electrochemistry to uncover the role of nitrogen in the biological reactivity of nitrogen-doped graphene†
Abstract
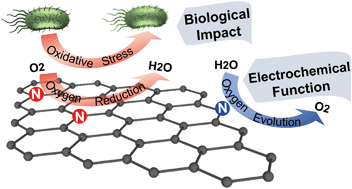
While nitrogen doping greatly broadens graphene applications, relatively little is known about the influence of this heteroatom on the biological activity of graphene. A set of systematically modified nitrogen-doped graphene (NG) materials was synthesized using the hydrothermal method in which the degree of N-doping and N-bonding type is manipulated using two nitrogen precursors (urea and uric acid) and different thermal annealing temperatures. The bioactivity of the NG samples was evaluated using the oxidation of the intracellular antioxidant glutathione (GSH) and bacterial viability (of Escherichia coli K12), and oxidative stress was identified as the predominant antibacterial mechanism. Two key energy-relevant electrochemical reactions, oxygen reduction reaction (ORR) and oxygen evolution reaction (OER), were used to characterize the influence of different N-types on the electronic properties of the NG materials. Electron-donating graphitic-N and electron-withdrawing pyridinic-N were identified as effective promoters for ORR and OER, respectively. The similar mechanisms between the GSH oxidation (indicative of oxidative stress) and ORR mechanisms reveal the role of graphitic-N as the active site in oxidative stress related bioactivity, independent of other consequential properties (e.g., defect density, surface area). This work advances a growing rational design paradigm for graphene family materials using chemical composition and further provides valuable insight into the performance-hazard tradeoffs of NG applications in related fields.


 Please wait while we load your content...
Please wait while we load your content...
