Comparing TiO2 nanoparticle formulations: stability and photoreactivity are key factors in acute toxicity to Daphnia magna†
Abstract
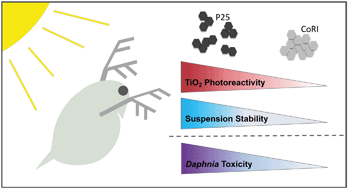
Given their widespread manufacture and use, it is anticipated that titanium dioxide nanoparticles (TiO2 NPs) will make their way into environmental surface waters where they may adversely impact biota. Characteristics of both the NPs themselves and the aquatic environment will determine the potential risks associated with release. While multiple water chemistries are often considered in the same study, differences in TiO2 NPs themselves have been overlooked. Photocatalytic reactive oxygen species generation will be governed by the intrinsic reactivity of a NP, the extent of aggregation in suspension, and the aquatic chemistry of the solution. Here, two different commercial formulations of TiO2 NPs are investigated in terms of aggregate stability, photoreactivity, and toxicity to the freshwater crustacean Daphnia magna. D. magna neonates were exposed to TiO2 NPs in 48 h acute toxicity tests under either visible or UV illumination, in either the presence or absence of natural organic matter (NOM). Negligible lethal toxicity was observed by either TiO2 formulation when illuminated by visible light, although swimming performance was significantly affected. For both NPs, the presence of UV light dramatically increased hydroxyl radical (˙OH) generation and toxicity. This toxicity was partially mitigated by the presence of NOM acting as a quenching agent for ˙OH. Differences in both aggregate stability and photoreactivity between the two TiO2 NPs are shown to influence toxicity. These results highlight that not all TiO2 should be treated equally, and that the particle stability and photoreactivity of an individual formulation must be taken into account when predicting risk.


 Please wait while we load your content...
Please wait while we load your content...