Heteroaggregation and dissolution of silver nanoparticles by iron oxide colloids under environmentally relevant conditions†
Abstract
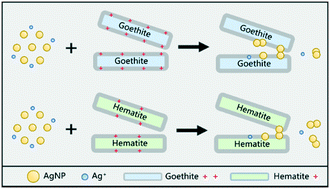
The ubiquity and abundance of iron oxides in the subsurface highlight their important roles in influencing the fate and transport of engineered silver nanoparticles (AgNPs). In this study, the adsorption behaviors of AgNPs on two naturally occurring iron oxides, goethite and hematite, were investigated under environmentally relevant conditions. The maximum surface coverage of AgNPs on iron oxides ranged between 0.014 and 0.326 mg m−2 depending on the investigated ionic strength and pH. The particle interactions (AgNPs–AgNPs and AgNPs–goethite/hematite) were probed by aggregation kinetics measurements using time-resolved dynamic light scattering and Derjaguin–Landau–Verwey–Overbeek theory calculations, which confirmed the predominant role of heteroaggregation in AgNP adsorption onto iron oxides. Multiple state-of-the-art characterization studies using X-ray absorption spectroscopy, attenuated total reflection-Fourier transform infrared spectroscopy, and X-ray diffraction substantiate the dominant electrostatic attractions between AgNPs and iron oxides. Moreover, AgNP dissolution was reduced in the presence of iron oxides. Goethite was more effective than hematite in retaining AgNPs (5.1 to 16.3-fold higher) and inhibiting AgNP dissolution (1.2 to 5.7-fold lower), due to their surface charge differences. Altogether, our findings provide compelling evidence of the dominant role played by electrostatic attractions in AgNP adsorption by iron oxides and of inhibition of AgNP dissolution during the heteroaggregation process, which has important implications for better evaluating the potential environmental impacts and risks of AgNPs in the iron oxide-rich subsurface.


 Please wait while we load your content...
Please wait while we load your content...