An alkaline polymer electrolyte CO2 electrolyzer operated with pure water†
Abstract
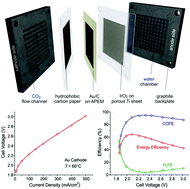
The electrochemical conversion of CO2 to fuels or chemicals is an attractive approach that can combine sustainable energy utilization and carbon recycling, but its practicability is still vague, because the CO2 reduction reaction (CO2RR) has hitherto been mostly studied in electrolyte solutions, with small current densities limited by the solubility of CO2. An important and urgent task is thus to develop CO2 electrolyzers that can demonstrate the feasibility of the large-scale conversion of CO2, in particular analogs of water electrolyzers using polymer electrolyte based gas-diffusion electrodes. Here we report a high-performance CO2 electrolyzer making use of alkaline polymer electrolytes (APEs). A highly conductive and stable APE, quaternary ammonia poly(N-methyl-piperidine-co-p-terphenyl) (QAPPT), was applied both as the membrane separator and the ionomer impregnating the gas-diffusion electrodes. Dry CO2 was fed into the Au cathode, with a faradaic efficiency of CO production (COFE) over 85%, and the oxygen evolution reaction (OER) occurred at the IrO2 anode fed with pure water. The cell was operated at temperatures up to 80 °C, and the current density reached 0.5 A cm−2 at 3 V at 60 °C. The stability of this electrolyzer was tested under 0.1 A cm−2, and the cell voltage remained stable at 2.25 V over 100 h, with the COFE staying at 90–95%.


 Please wait while we load your content...
Please wait while we load your content...