Mixed lead carboxylates relevant to soap formation in oil and tempera paintings: the study of the crystal structure by complementary XRPD and ssNMR†
Abstract
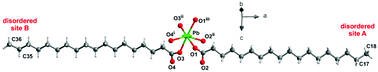
Long-chain lead carboxylates, on the one hand, represent compounds for versatile industrial applications in high-tech industries, while on the other hand, they are predominant constituents of secondary products of saponification of paint layers in works of art. Affecting significantly the appearance and stability of painted works of art, saponification is one of the most serious problems of preservation of cultural heritage objects. Despite their versatility as well as hazardousness, there is a paucity of single-crystal X-ray structures of long-chain carboxylates, due to difficulties in preparing single crystals of sufficient quality. We studied the crystal structure of polycrystalline mixed lead carboxylates of the formula Pb(C16)2−x(C18)x (x = 0; 0.25; 0.5; 0.75; 1; 1.5; 2), where C16 and C18 stand for hexadecanoate (palmitate) and octadecanoate (stearate) anions, respectively, by complementary X-ray powder diffraction (XRPD) and 13C and 207Pb solid state NMR (ssNMR). Mixed lead carboxylates consisting of hexadecanoate and octadecanoate are relevant to the formation of soaps in egg yolk and/or oil-based binders combined with lead-based pigments, which belong to the most common pigments in history. Combining an advanced XRPD analysis with a comparative analysis of ssNMR parameters, we described the structural model of mixed lead carboxylates. We revealed that both hexadecanoate (C16) and octadecanoate (C18) chains are present in one crystal structure, creating the statistical disorder at the ethyl end of the chains. Based on the 207Pb ssNMR spectra, we revealed two distinct local environments of lead atoms, corresponding to the symmetrically (i.e., (C16)–Pb–(C16) and/or (C18)–Pb–(C18) and asymmetrically (i.e., (C16)–Pb–(C18)) substituted lead carboxylates, and we confirmed the formation of a holo-directed structure for both the structural motifs. The structural models were applied to identify the neo-formed crystalline lead soap in a model experiment simulating the simplified historic paint consisting of the pigment lead tin yellow type I and emulsion binder prepared from egg yolk and linseed oil. We identified the secondary product as a mixed lead carboxylate of the composition Pb(C16)(C18).


 Please wait while we load your content...
Please wait while we load your content...