Influence of ancillary ligands and solvents on the nuclearity of Ni–Ln complexes†
Abstract
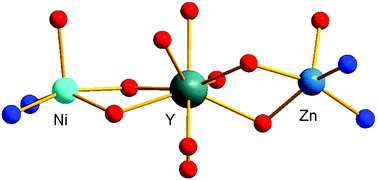
A Schiff base ligand resulting from the reaction of ovanillin and 2,2-dimethyl-1,3-diaminopropane allows the preparation of hetero-dinuclear [Ni–Ln]3+ or -trinuclear [Ni–Ln–Ni]3+ complexes. Although empirical parameters for rationalizing the strength of the ferromagnetic Ni–Gd interaction have already been discussed in several papers, no systematic study has been devoted to the control of the nuclearity of such complexes. With the help of structural determinations, we demonstrate the role of solvent and of the nature of ancillary ligands, linked to the Ln ions, in nuclearity. For instance, the presence of one chelating nitrato ligand is already sufficient to impede an increase in the nuclearity, while the replacement of nitrato ligands by chloride anions still yields dinuclear Ni–Ln complexes. This experimental result evidences the role of protic solvents. In contrast, the use of lanthanide salts, soluble in non-protic solvents, allows the isolation of dinuclear [Ni–Ln]3+ or trinuclear cationic [LNi–Ln–NiL]3+ complexes, depending on the Ni/Ln ratio. A further synthetic step can be overtaken by the reaction of a Ni–Ln complex, soluble in a non-protic solvent, with a LM complex (M = Cu, Zn). By doing so, a heterotrinuclear complex made of three different metal ions, two distinct 3d ions and a 4f one, has been isolated and structurally characterized. Note that the Ni coordination number decreases from 6 to 5 on going from the dinuclear complex to the trinuclear one. Also, the replacement of water molecules by chloride ligands in the hexacoordinate Ni complexes induces a net increase of the positive zero-field splitting parameter D to 20 cm−1, which is supported by ab initio calculations. Although the Ni–Ln (Ln = Gd, Tb, Dy) magnetic interactions are ferromagnetic, the corresponding trinuclear complexes are devoid of SMM properties in the absence of an applied magnetic field.


 Please wait while we load your content...
Please wait while we load your content...