Directing self-assembly in solution towards improved cooperativity in Fe(iii) complexes with amphiphilic tridentate ligands†
Abstract
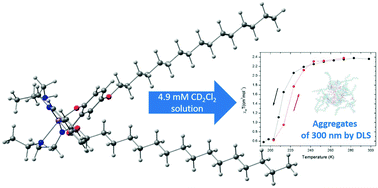
An amphiphilic iron(III) complex with a tridentate Schiff-base ligand was prepared by condensation of a hexadecyloxy functionalised salycylaldehyde with a diamine followed by complexation with FeCl2 and anion methathesis with NaClO4. The complex shows spin crossover both in the solid state and solution. However in solution self-assembly and consequently aggregation of individual molecules form concentration dependent particles with sizes of 300 nm for higher concentrations, or 5 nm for lower concentrations. Aggregate formation was confirmed by NANO-flex 180° DLS Size, scan-rate dependent cyclic voltammetry and scanning electron microscopy. Molecular simulations were used to investigate the self-assembly of the complex in solution, including the role of residual water molecules. The simulations showed the self-assembly of reverse micelle-like structures when a small water cluster is inserted in solution, whereas no large aggregates formed in dehydrated environments. The perchlorate anions were found near the metal centres, stabilizing the aggregates around the water pool. Simulations of pre-assembled structures further showed the lack of stability of large aggregates in the absence of water. The larger aggregates promoted efficient communication between the iron(III) centres and the compound displayed spin crossover in solution at around 220 K with a 10 K hysteresis window, as measured by NMR and SQUID magnetometry.
- This article is part of the themed collection: Nitrogen Ligands


 Please wait while we load your content...
Please wait while we load your content...